FDA: Philippines struggling to secure COVID-19 vaccines | ABS-CBN

Welcome, Kapamilya! We use cookies to improve your browsing experience. Continuing to use this site means you agree to our use of cookies. Tell me more!
FDA: Philippines struggling to secure COVID-19 vaccines
FDA: Philippines struggling to secure COVID-19 vaccines
ABS-CBN News
Published Apr 16, 2021 08:14 AM PHT
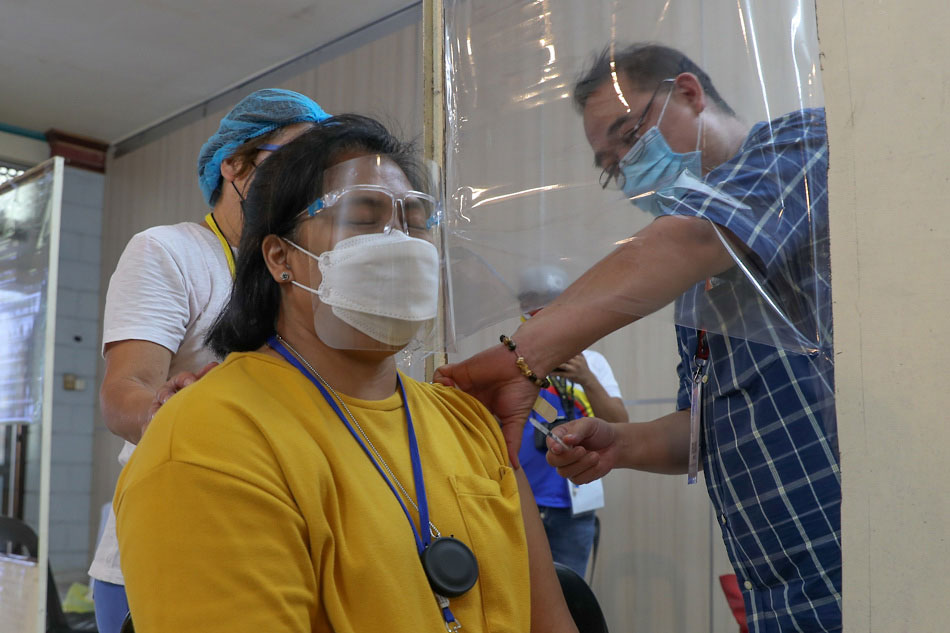
MANILA—The Philippines is struggling to secure enough COVID-19 vaccines for its entire population, as coronavirus infections surpassed the 900,000-mark, the country's drug regulator said Friday.
MANILA—The Philippines is struggling to secure enough COVID-19 vaccines for its entire population, as coronavirus infections surpassed the 900,000-mark, the country's drug regulator said Friday.
"So far, nahihirapan pong kumuha ng supply 'yong gobyerno (the government is struggling to secure supplies)," Food and Drug Administration director-general Eric Domingo told Teleradyo.
"So far, nahihirapan pong kumuha ng supply 'yong gobyerno (the government is struggling to secure supplies)," Food and Drug Administration director-general Eric Domingo told Teleradyo.
Data from the Department of Health, as of April 13, showed that the country has administered over 1.2 million doses. The Philippines began its inoculation drive on March 1.
Data from the Department of Health, as of April 13, showed that the country has administered over 1.2 million doses. The Philippines began its inoculation drive on March 1.
Domingo said his office was still evaluating the applications for emergency use of jabs developed by India's Bharat Biotech and Belgium-based Janssen Pharmaceuticals.
Domingo said his office was still evaluating the applications for emergency use of jabs developed by India's Bharat Biotech and Belgium-based Janssen Pharmaceuticals.
ADVERTISEMENT
US-based company Moderna has also yet to submit its EUA application to FDA, he added.
US-based company Moderna has also yet to submit its EUA application to FDA, he added.
"Matagal na po silang (Moderna) nagpaparamdam pero hindi pa po naga-apply. Pati po kami nakaabang na rin do'n sa kanilang appliation," Domingo said.
"Matagal na po silang (Moderna) nagpaparamdam pero hindi pa po naga-apply. Pati po kami nakaabang na rin do'n sa kanilang appliation," Domingo said.
(They have been putting out feelers for some time but has yet to apply. We are also waiting for their application.)
(They have been putting out feelers for some time but has yet to apply. We are also waiting for their application.)
In a taped public address on Thursday night, President Rodrigo Duterte said industrial countries "are not ready really to let go of their stocks of vaccine."
In a taped public address on Thursday night, President Rodrigo Duterte said industrial countries "are not ready really to let go of their stocks of vaccine."
"When will have that stock sufficient to vaccinate the people? I really do not know. Nobody knows," Duterte said.
"When will have that stock sufficient to vaccinate the people? I really do not know. Nobody knows," Duterte said.
ADVERTISEMENT
"There's no sufficient supply to inoculate the world. Matagal pa 'to. Sabihin ko sa 'yo marami pang mamamatay dito."
"There's no sufficient supply to inoculate the world. Matagal pa 'to. Sabihin ko sa 'yo marami pang mamamatay dito."
On Thursday, the global Covax scheme launched a push for an extra $2 billion in donations, saying it needed the money in advance to reserve COVID-19 vaccine doses.
On Thursday, the global Covax scheme launched a push for an extra $2 billion in donations, saying it needed the money in advance to reserve COVID-19 vaccine doses.
The Covax facility ensures the 92 poorest participating economies can access coronavirus jabs, with the cost covered by donors.
The Covax facility ensures the 92 poorest participating economies can access coronavirus jabs, with the cost covered by donors.
The Philippines is a recipient of the Covax program.
The Philippines is a recipient of the Covax program.
To date, only 4 anti-coronavirus shots have obtained regulatory approval in the country. They are Pfizer-BioNTech, AstraZeneca-Oxford, Sinovac and Gamaleya Institute.
To date, only 4 anti-coronavirus shots have obtained regulatory approval in the country. They are Pfizer-BioNTech, AstraZeneca-Oxford, Sinovac and Gamaleya Institute.
ADVERTISEMENT
On Thursday, the FDA recommended that the country resume its rollout of the AstraZeneca vaccine to individuals aged below 60 despite reports abroad of potential links to blood clots.
On Thursday, the FDA recommended that the country resume its rollout of the AstraZeneca vaccine to individuals aged below 60 despite reports abroad of potential links to blood clots.
Domingo on Friday said the FDA sought the guidance of the World Health Organization and local experts, and found out the jabs' advantages outweighed the risks.
Domingo on Friday said the FDA sought the guidance of the World Health Organization and local experts, and found out the jabs' advantages outweighed the risks.
"Nakita natin na very rare itong blood clotting. Sa ibang lugar, its 1 for every 1 million. No'ng kuwenenta natin, talagang mas malaki ang risk na magkaroon ng COVID at mamatay sa COVID," he said.
"Nakita natin na very rare itong blood clotting. Sa ibang lugar, its 1 for every 1 million. No'ng kuwenenta natin, talagang mas malaki ang risk na magkaroon ng COVID at mamatay sa COVID," he said.
(We saw that blood clotting was rare. In other countries, its 1 for every 1 million. The risk of catching and dying from COVID-19 is greater.)
(We saw that blood clotting was rare. In other countries, its 1 for every 1 million. The risk of catching and dying from COVID-19 is greater.)
The AstraZeneca vaccine uses an adenovirus, a harmless class of common-cold viruses, to introduce coronavirus proteins into cells in the body and trigger an immune response.
The AstraZeneca vaccine uses an adenovirus, a harmless class of common-cold viruses, to introduce coronavirus proteins into cells in the body and trigger an immune response.
ADVERTISEMENT
The Philippines has received 525,600 AstraZeneca doses, almost all of which have been administered. The country's next delivery of the UK-developed vaccine might arrive in May, Domingo said last week.
The Philippines has received 525,600 AstraZeneca doses, almost all of which have been administered. The country's next delivery of the UK-developed vaccine might arrive in May, Domingo said last week.
It is 1 of only 2 vaccine brands currently available in the Philippines, the other being China's Sinovac.
It is 1 of only 2 vaccine brands currently available in the Philippines, the other being China's Sinovac.
RELATED VIDEO
Read More:
Teleradyo
Sakto
Food and Drug Administration
FDA
Eric Doming
COVID-19
coronavirus
COVID-19 vaccines
AstraZeneca COVID-19 vaccine
ADVERTISEMENT
ADVERTISEMENT