'Hindi pa napag-aralan': FDA has not yet evaluated Chinese COVID-19 jab used on Duterte | ABS-CBN

Welcome, Kapamilya! We use cookies to improve your browsing experience. Continuing to use this site means you agree to our use of cookies. Tell me more!
'Hindi pa napag-aralan': FDA has not yet evaluated Chinese COVID-19 jab used on Duterte
'Hindi pa napag-aralan': FDA has not yet evaluated Chinese COVID-19 jab used on Duterte
ABS-CBN News
Published May 04, 2021 10:16 AM PHT
|
Updated May 04, 2021 10:33 AM PHT
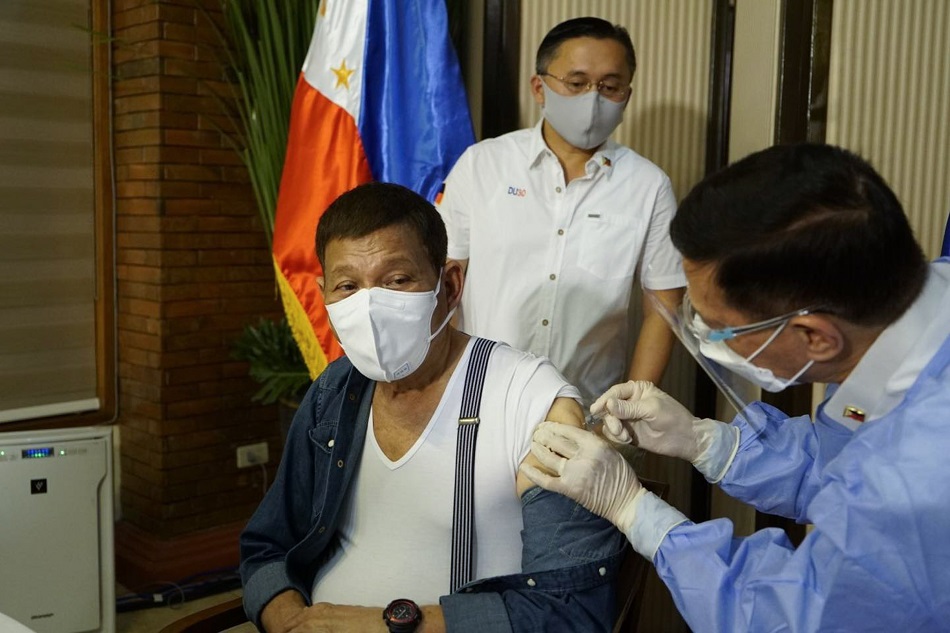
MANILA - The Food and Drug Administration has yet to evaluate the Chinese vaccine that was used to inoculate President Rodrigo Duterte against COVID-19, its chief said Tuesday.
MANILA - The Food and Drug Administration has yet to evaluate the Chinese vaccine that was used to inoculate President Rodrigo Duterte against COVID-19, its chief said Tuesday.
The FDA has yet to issue an emergency use authorization for Sinopharm's COVID-19 vaccine, said director general Eric Domingo. It only granted compassionate special permit for 10,000 doses of the Chinese vaccine to be used by the Presidential Security Group, he added.
The FDA has yet to issue an emergency use authorization for Sinopharm's COVID-19 vaccine, said director general Eric Domingo. It only granted compassionate special permit for 10,000 doses of the Chinese vaccine to be used by the Presidential Security Group, he added.
"I think it’s a personal preference. Sa amin po, ang Sinopharm hindi pa napag-aralan ng FDA 'yan," he told ABS-CBN's Teleradyo on why the President chose to be inoculated with the Chinese vaccine.
"I think it’s a personal preference. Sa amin po, ang Sinopharm hindi pa napag-aralan ng FDA 'yan," he told ABS-CBN's Teleradyo on why the President chose to be inoculated with the Chinese vaccine.
(The FDA has yet to study the Sinopharm vaccine.)
(The FDA has yet to study the Sinopharm vaccine.)
ADVERTISEMENT
"'Pag sinabing compassionate special permit, hindi po 'yun authorization na binigay ng FDA. In this case, yun pong head ng PSG hospital siya ang naga-guarantee na inaral niya ang bakuna and they take full responsibility for it. Sa amin po dito sa FDA di pa po namin na-evaluate ang bakunang iyan."
"'Pag sinabing compassionate special permit, hindi po 'yun authorization na binigay ng FDA. In this case, yun pong head ng PSG hospital siya ang naga-guarantee na inaral niya ang bakuna and they take full responsibility for it. Sa amin po dito sa FDA di pa po namin na-evaluate ang bakunang iyan."
(When you say compassionate special permit, it's not an authorization given by the FDA. In this case, the head of the PSG hospital guarantees they studied the vaccine and they take full responsibility for it. Here at the FDA, we have yet to evaluate the vaccine.)
(When you say compassionate special permit, it's not an authorization given by the FDA. In this case, the head of the PSG hospital guarantees they studied the vaccine and they take full responsibility for it. Here at the FDA, we have yet to evaluate the vaccine.)
The World Health Organization has an interim evaluation of Sinopharm's jab which had similarities with Sinovac's vaccine, Domingo said.
The World Health Organization has an interim evaluation of Sinopharm's jab which had similarities with Sinovac's vaccine, Domingo said.
"Unang-una safe ang vaccine kasi inactivated virus katulad ng Sinovac...Nakita rin po na maganda ang efficacy," he said.
"Unang-una safe ang vaccine kasi inactivated virus katulad ng Sinovac...Nakita rin po na maganda ang efficacy," he said.
(First of all, it's safe because it uses inactivated virus like Sinovac...It also found the efficacy rate was good.)
(First of all, it's safe because it uses inactivated virus like Sinovac...It also found the efficacy rate was good.)
"Continuing pa rin ang evaluation nila for this vaccine pero so far maganda naman po ang nakikitang mga resulta."
"Continuing pa rin ang evaluation nila for this vaccine pero so far maganda naman po ang nakikitang mga resulta."
(Their evaluation for this vaccine continues but so far they're seeing good results.)
(Their evaluation for this vaccine continues but so far they're seeing good results.)
Some 2,000 to 3,000 soldiers guarding the President are set to receive the vaccine while the remaining doses will be given to their wives, Domingo said.
Some 2,000 to 3,000 soldiers guarding the President are set to receive the vaccine while the remaining doses will be given to their wives, Domingo said.
Read More:
Teleradyo
Rodrigo Duterte
Sinopharm
FDA
Food and Drug Administration
Eric Domingo
COVID-19 vaccine
Philippines COVID-19 vaccination
COVID-19
coronavirus
ADVERTISEMENT
ADVERTISEMENT