Pfizer eyes COVID vaccine for all variants before 2023 | ABS-CBN
ADVERTISEMENT

Welcome, Kapamilya! We use cookies to improve your browsing experience. Continuing to use this site means you agree to our use of cookies. Tell me more!
Pfizer eyes COVID vaccine for all variants before 2023
Pfizer eyes COVID vaccine for all variants before 2023
Robin Millard,
Agence France-Presse
Published Apr 15, 2022 08:17 AM PHT
GENEVA - A COVID-19 vaccine effective against multiple variants is possible before the end of 2022, the head of US pharmaceutical giant Pfizer said Wednesday.
GENEVA - A COVID-19 vaccine effective against multiple variants is possible before the end of 2022, the head of US pharmaceutical giant Pfizer said Wednesday.
Chairman Albert Bourla said the firm was also working on producing a vaccine that could provide good protection for a whole year, meaning people would come back annually for boosters, as with influenza shots.
Chairman Albert Bourla said the firm was also working on producing a vaccine that could provide good protection for a whole year, meaning people would come back annually for boosters, as with influenza shots.
"I hope, clearly by autumn... that we could have a vaccine" that worked against not only the dominant Omicron but all known variants, he said.
"I hope, clearly by autumn... that we could have a vaccine" that worked against not only the dominant Omicron but all known variants, he said.
"It is a possibility that we have it by then. It is not a certainty," he told a media briefing organised by the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA) big pharma lobby group.
"It is a possibility that we have it by then. It is not a certainty," he told a media briefing organised by the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA) big pharma lobby group.
ADVERTISEMENT
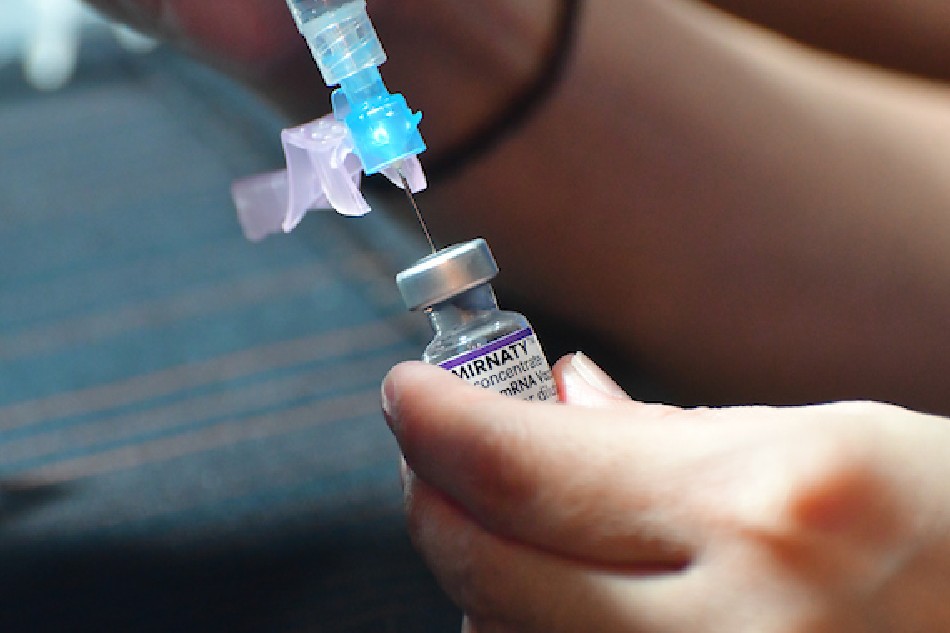
The Pfizer-BioNTech vaccine is one of the most effective against COVID-19.
The Pfizer-BioNTech vaccine is one of the most effective against COVID-19.
Though, like other jabs, it has seen waning effectiveness against the now-dominant Omicron variant of the virus, it still offers strong protection against serious disease, hospitalization and death.
Though, like other jabs, it has seen waning effectiveness against the now-dominant Omicron variant of the virus, it still offers strong protection against serious disease, hospitalization and death.
IFPMA director general Thomas Cueni said the world had to learn to live with the virus that causes COVID-19 disease, saying: "We are beyond the time when you can eradicate SARS-CoV-2."
IFPMA director general Thomas Cueni said the world had to learn to live with the virus that causes COVID-19 disease, saying: "We are beyond the time when you can eradicate SARS-CoV-2."
Bourla said there was a risk of vaccine fatigue, predicting that few people who have so far declined the chance to be vaccinated would change their minds, and suggesting that fewer people would come back for fourth doses than came forward for a third, booster dose.
Bourla said there was a risk of vaccine fatigue, predicting that few people who have so far declined the chance to be vaccinated would change their minds, and suggesting that fewer people would come back for fourth doses than came forward for a third, booster dose.
"What the world really needs is a vaccine that will last a year. I think this is what will become the optimal public health solution," he said.
"What the world really needs is a vaccine that will last a year. I think this is what will become the optimal public health solution," he said.
ADVERTISEMENT
"It's way more easy to be administered and have the population be compliant with that.
"It's way more easy to be administered and have the population be compliant with that.
"It is very challenging, technically, to do it with this virus -- but we are working on it."
"It is very challenging, technically, to do it with this virus -- but we are working on it."
IP waiver plan blasted
The World Health Organization is aware of 153 COVID-19 vaccines that have been in clinical development -- tested on humans -- and 196 in pre-clinical development.
The World Health Organization is aware of 153 COVID-19 vaccines that have been in clinical development -- tested on humans -- and 196 in pre-clinical development.
But so far, the UN health agency has only authorised eight vaccines and versions thereof: those made by Pfizer-BioNTech, AstraZeneca, Janssen, Moderna, Sinovac, Sinopharm, Bharat Biotech and Novavax.
But so far, the UN health agency has only authorised eight vaccines and versions thereof: those made by Pfizer-BioNTech, AstraZeneca, Janssen, Moderna, Sinovac, Sinopharm, Bharat Biotech and Novavax.
The IFPMA briefing condemned suggestions that intellectual property rights on COVID-19 vaccines should be temporarily lifted during the pandemic, in a bid to increase production.
The IFPMA briefing condemned suggestions that intellectual property rights on COVID-19 vaccines should be temporarily lifted during the pandemic, in a bid to increase production.
ADVERTISEMENT
"I'm stunned that the proposed IP waiver is still debated while supplies are far outstripping demand," said Cueni.
"I'm stunned that the proposed IP waiver is still debated while supplies are far outstripping demand," said Cueni.
With more than a billion vaccine doses being produced every month, Eli Lilly chairman David Ricks called it "a solution looking for a problem", while Bourla branded the idea "insane".
With more than a billion vaccine doses being produced every month, Eli Lilly chairman David Ricks called it "a solution looking for a problem", while Bourla branded the idea "insane".
In a later WHO press conference, the UN health agency's chief scientist Soumya Swaminathan said the vaccine supply situation had dramatically improved in recent months.
In a later WHO press conference, the UN health agency's chief scientist Soumya Swaminathan said the vaccine supply situation had dramatically improved in recent months.
The problem, she said, was more one of logistics and getting doses into arms in countries, especially in Africa, where only 13 percent have been fully jabbed.
The problem, she said, was more one of logistics and getting doses into arms in countries, especially in Africa, where only 13 percent have been fully jabbed.
"We are able to now supply as many doses as countries want, on demand, to them. The issue now really is on delivery," she said.
"We are able to now supply as many doses as countries want, on demand, to them. The issue now really is on delivery," she said.
© Agence France-Presse
RELATED VIDEO
ADVERTISEMENT
ADVERTISEMENT